 
People who receive a COVID-19 vaccination can register with v-safe to voluntarily report side effects, such as pain or nausea, for the first days, weeks, and months after injection. The v-safe after vaccination health checker was created by Oracle developers for the US Centers for Disease Control and Prevention (CDC), using the Oracle APEX low-code development tool, Oracle Analytics, and Oracle Cloud Infrastructure. 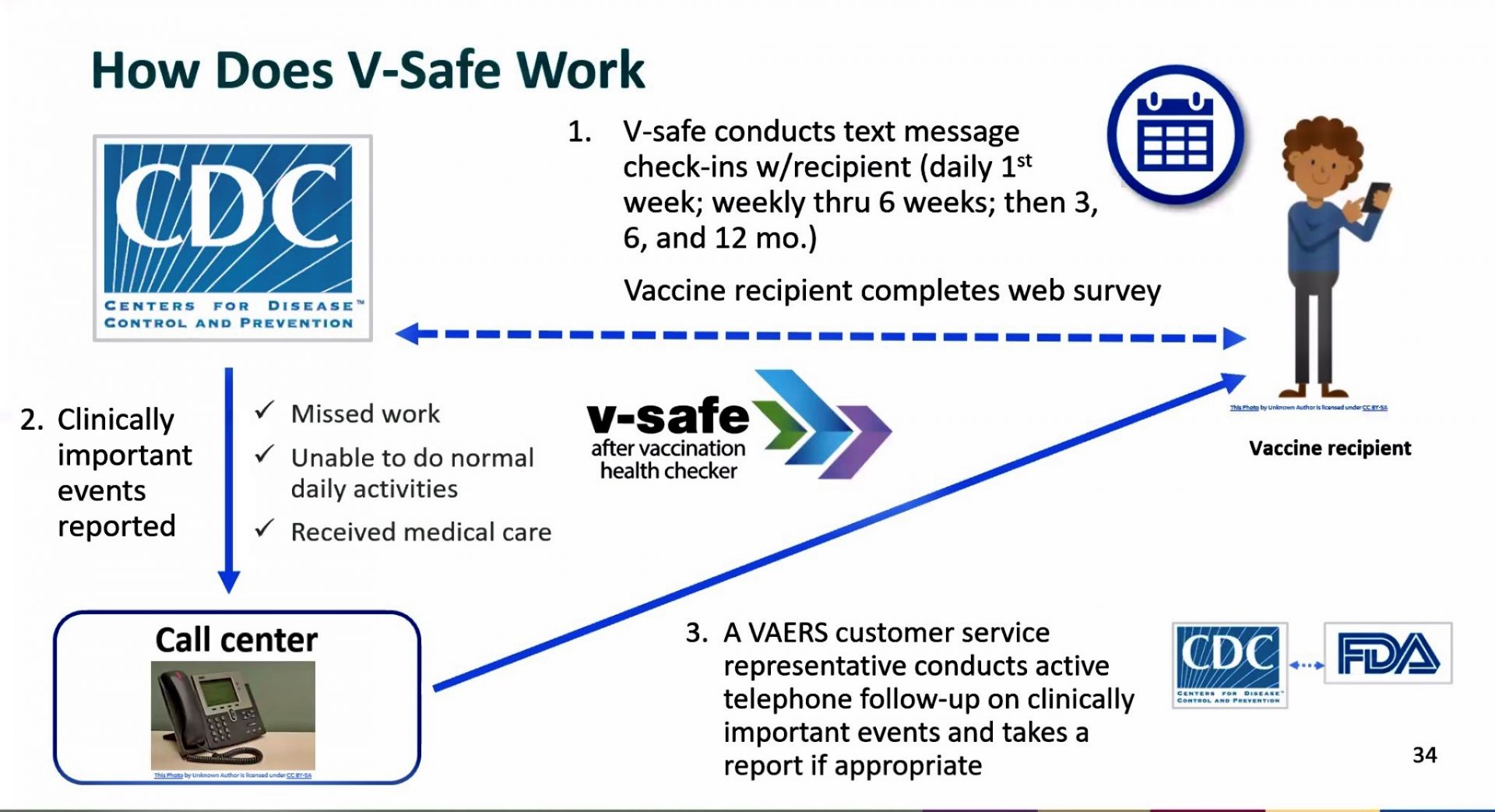
A mobile solution called v-safe sits at the heart of that effort in the United States. The biggest examples of this revolution in health science and data collection come directly from COVID-19, with the massive effort to collect real-world data to monitor the safety and efficacy of the vaccines that the scientific community so quickly created, tested, and brought to market. Henry McNamara, SVP and General Manager, Oracle Health Sciences “Since the pandemic didn't just last for three months, they're starting to realize that not only was this a mitigation strategy, that this is actually a better way of doing the work.” “Researchers had to pause and think, and they've come up with new ways to conduct clinical development,” says Katherine Vandebelt, global vice president of innovation for Oracle Health Sciences. Those new techniques are likely to stick well past the pandemic. 
And two, those that tried to keep running needed to figure out how to operate when patients couldn’t come into offices or health centers for their trial visits.Ĭlinical trial operators needed new, decentralized data collection models, tapping into telemedicine, digitized source data (eSource), and electronic consent forms (eConsent). One, pharmaceutical companies and researchers turned their attention to treatments and vaccines for COVID-19. In March of 2020, more than 50% of the clinical trials running at the time were shut down, for two main reasons. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
